20. At the bottom of an old kettle you might notice some white residue. This is due to calcium deposits from the water in your tap. What is water that is infused with many calcium ions called?
From Quiz The Dark Side of Calcium
Answer:
Hard water
High concentrations of both calcium and magnesium ions dissolved in water is more commonly known as 'hard water'. At home, this will leave white residue on your taps, kettles, shower heads, and any other devices that use water, such as, more often than not, heating elements. As a quick test to see how hard your water is, pour some soap into tap water in a bowl. The easier it is to make suds, the softer your water is.
In Canada, Regina, Saskatchewan has one of the hardest tap waters, whereas Vancouver, British Columbia has one of the softest.
As far as health concerns and hard water go, there haven't been any conclusive papers published that link hard water to any diseases and conditions. So, the only real trouble with hard water is cleaning the white crud out of the pipes or off of surfaces. Usually, vinegar is an easy way to clean it off.
 If you've ever wanted to learn about antimony-or if you're eager to show off your deep knowledge of it-here's the quiz you've been waiting for your whole life (sorry). From its symbol to its uses in plastic and batteries, this one runs the gamut. Enjoy!
If you've ever wanted to learn about antimony-or if you're eager to show off your deep knowledge of it-here's the quiz you've been waiting for your whole life (sorry). From its symbol to its uses in plastic and batteries, this one runs the gamut. Enjoy! 





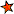

 Quick Question
Quick Question