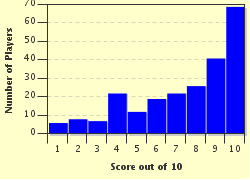
Quiz Answer Key and Fun Facts
1. Organic chemistry is the study of the molecules which, among other things, form the basis of life. Which element is the structural basis of all the molecules which are included in the field of organic chemistry?
2. The simplest organic molecules contain only two elements. The very smallest organic molecule is methane, which consists of molecules that have a single carbon atom and four atoms of what other element?
3. You have probably heard about saturated and unsaturated fats in relation to dietary issues. The term unsaturated refers to molecules which have one or more double (or triple) bonds between atoms of which element?
4. To keep peanut butter from separating and forming an oily layer as it stands on the shelf, it used to be common practice to reduce the amount of unsaturated fats in the oil being used. The atoms of what element were added to increase the proportion of saturated molecules in the oil?
5. A group of organic molecules that we encounter regularly in our food is carbohydrates. Which of these elements is NOT found in a carbohydrate molecule?
6. The well-loved chemical ethanol (also known as drinking alcohol) contains a functional group called hydroxyl. What atom in the hydroxyl group is bonded onto one of the two carbon atoms that forms the main chain of this molecule?
7. When an alcohol is oxidised, it typically forms a carbonyl group. A carbonyl group has a double bond between a carbon atom and another atom of which type?
8. When an alcohol is oxidised to form an aldehyde or a ketone, what kind of atom is removed from the molecule during the reaction?
9. When a primary alcohol is completely oxidised, it becomes an acid. The ethanol in wine, for example, can be oxidised to produce vinegar, formally known as ethanoic (or acetic) acid. When an organic acid dissolves in water, what kind of ion is formed?
10. The difference between an amino acid and an ordinary organic acid is that an amino acid includes which of the following kinds of atom?
Source: Author
looney_tunes
This quiz was reviewed by FunTrivia editor
WesleyCrusher before going online.
Any errors found in FunTrivia content are routinely corrected through our feedback system.